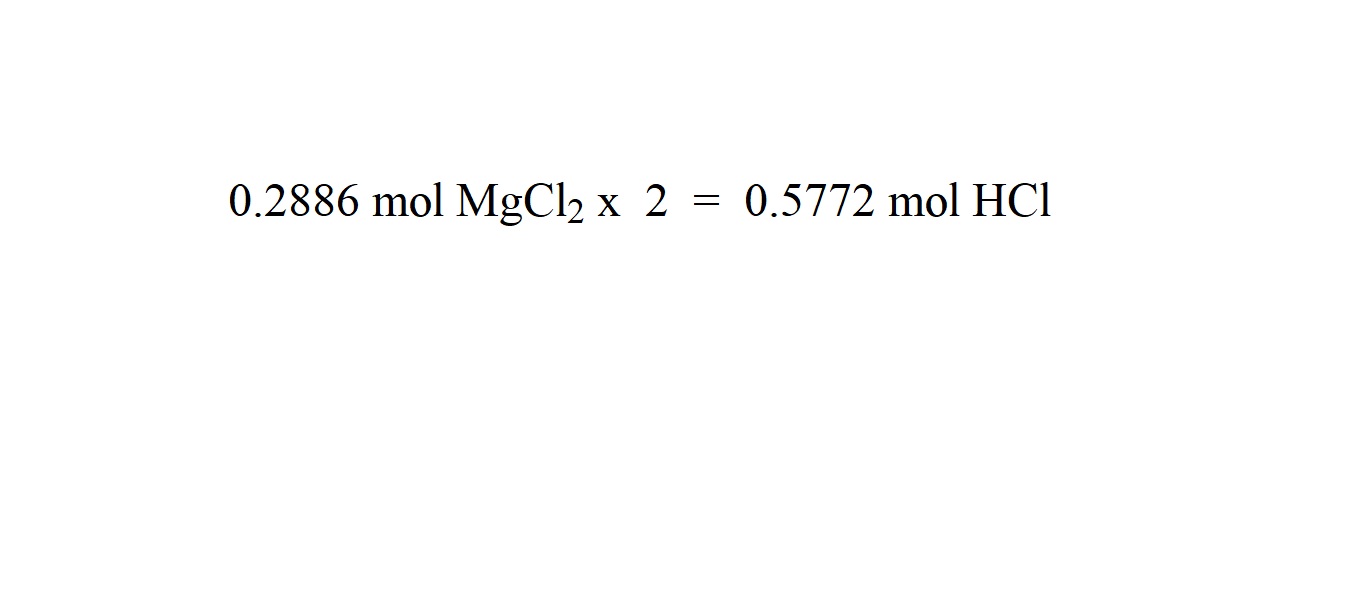
Note: Sodium hydroxide is a highly hazardous chemical that must be handled cautiously. The resulting solution should be clear and colorless. 
Mix until all the sodium hydroxide dissolves in water. Be careful when adding water, as the reaction between sodium hydroxide and water is highly exothermic and produces a lot of heat. Slowly add distilled or deionized water to the container while stirring with a glass rod. 
Take the measured amount of sodium hydroxide in a dry and clean glass beaker. For example, to prepare a 1 M sodium hydroxide solution, 40 grams of sodium hydroxide are required for every liter of water. Take the required amount of sodium hydroxide. Protective equipment, such as gloves and gogglesīefore using sodium hydroxide, wear all protective equipment, such as gloves and goggles, to avoid injury. Glass beaker or another suitable container The steps for preparing sodium hydroxide solution are as follows: Materials Required NaOH solution is prepared by dissolving pellets or flakes of solid sodium hydroxide in water. And it is used in the petroleum industry to refine crude oil as well as in the production of various chemicals such as plastics and pharmaceuticals. Sodium hydroxide solution is primarily used in a wide range of industrial and laboratory applications, such as in the manufacture of soaps, detergents, paper, and textiles. Sodium hydroxide solution, also called caustic soda solution, sodium hydroxide is a strong base, and its solution is highly alkaline (or alkaline). When it is heated above its melting point, sodium hydroxide melts and becomes liquid. It is solid at room temperature and is usually found in the form of pellets or flakes. The melting point of sodium hydroxide is 318.4 ☌ (605.1 ☏) at standard atmospheric pressure. The density of other concentrations will be different. The density of a 50% sodium hydroxide solution is approximately 1.525 g/mL at room temperature.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
